Showing posts with label ALXN. Show all posts
Showing posts with label ALXN. Show all posts
Monday, December 14, 2020
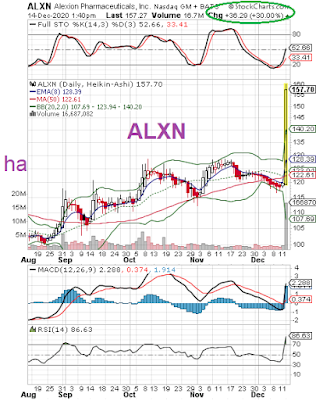
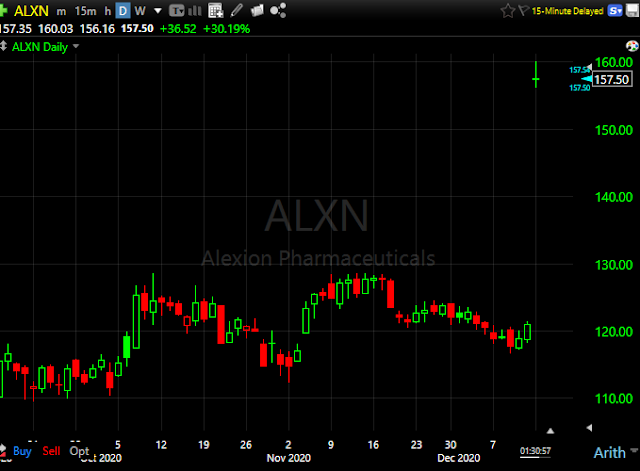
Alexion Pharma (ALXN) to be acquired by AstraZeneca (AZN) for $175 per share
Labels:
ALXN,
AZN,
mergers & acquisitions
Friday, May 8, 2020
Portola (PTLA) to be acquired by Alexion (ALXN) for $18 /sh
Alexion Pharmaceuticals announced Tuesday that it would spend $1.4 billion to acquire Portola Pharmaceuticals, which sells a medicine called Andexxa used to stop life-threatening bleeding in patients who have been given certain anticoagulants.
Labels:
ALXN,
mergers & acquisitions,
PTLA
Wednesday, October 16, 2019
Achillion Pharma (ACHN) to be acquired by Alexion (ALXN)
Achillion Pharma to be acquired by Alexion (ALXN) for $6.30/share in cash + CVRs worth up to $2.00/share
To be acquired by Alexion (ALXN) for an all-cash initial consideration of approx. $930 mln, or $6.30/share; potential additional contingent considerations, which hinge on clinical and regulatory developments such as FDA approval of danicopan and ACH-5228 Phase 3 initiation, could elevate the amount payable to co's shareholders to $8.30/share. Alexion looks forward to applying its "development expertise" to co's oral Factor D inhibitors in an endeavor to unlock the approach's potential and deliver benefits to patients. The stock moves to its highest levels in almost three years.
** ACHN **
Labels:
ACHN,
ALXN,
mergers & acquisitions
Monday, February 4, 2019
Alexion Pharma (ALXN) reported earnings on Mon 4 Feb 2019 (b/o)
** charts after earnings **
Alexion Pharma beats by $0.30, beats on revs; guides FY19 EPS above consensus, revs below consensus
- Reports Q4 (Dec) earnings of $2.14 per share, excluding non-recurring items, $0.30 better than the S&P Capital IQ Consensus of $1.84; revenues rose 24.1% year/year to $1.13 bln vs the $1.06 bln S&P Capital IQ Consensus.
- SOLIRIS net product sales were $976.7 million, compared to $791.9 million in the fourth quarter of 2017, representing a 23 percent increase. SOLIRIS volume increased 28 percent year-over-year.
- Co issues guidance for FY19, sees EPS of $9.10-9.30 vs. $8.80 S&P Capital IQ Consensus; sees FY19 revs of $4.625-4.7 bln vs. $4.75 bln S&P Capital IQ Consensus.
Earnings this week : Feb. 4 - 8, 2019 (wk 6)
Earnings expected this week:
Monday (Feb 4)
Tuesday (Feb 5)
Thursday (Feb 7)
Friday (Feb 8)
Monday (Feb 4)
Tuesday (Feb 5)
- Morning: ADM AME ARMK BDX CHD CNC EL EMR ENR INGR IT LII LITE RL
- Afternoon: ALL APC APU CB CERN DATA DIS DOX EA MANH MCHP MODN MTSI MYGN MXL OI PAA PAYC SAVE SCSC SFLY SNAP SWKS TCS TENB ULTI USNA VNOM VRTX ZEN
- Morning: ABG BSX BCO CSTE CPRI CTSH CMI ELAN FDC FDV GM HUM LLY NYT REGN SPOT TRVG TTWO
- Afternoon: ACLS AEL AHL AINV AOSL BLKB CCK CCMP CDAY CENT CINF CMG CSGS CUB CUZ DHT ECHO EGP ELY ENS ENSG FEYE FLO FLT FORM FTNT GBDC GPRO HQY ICHR IRBT KNL LCI LNC MC MET MTCH MTRX NEWR NOV NTGR NTR NXPI ORLY OSUR PAHC PCTY PRU PSDO PSEC RGLD RYN SONO TRMB TTGT TTMI TWO UHAL VVV WERN ZNGA
Thursday (Feb 7)
- Morning: ADNT ADS AMAG ANDX ARW BCE BR BSIG BV CAH CDW CEIX CNHI COR DNKN DTE EIGI EPC FCAU GRA GRUB HAIN HBI ICE K LABL LCII LH MAC MAS MD MMS MPC MPLX MPW ODFL PAG PBH PENN PM PRLB PTEN SEE SNA SNY SPB SPGI TGI TKR TMUS TPR TSN TWTR VIRT VSTO WLTW WMS WWE YUM
- Afternoon: ATEN ATGE ALNY ASYS ANGI MT ATHN BECN BHE BKH CARB CSL CBL CCS COLM OFC DXC EGAN EHC ESE EEFT EXPE FISV FSCT FTV FET FWRD G HUBG IAC KN LGND MTW MCFT MAT MTD MOBL MHK MSI NCR UEPS NR NWSA NUAN OMCL CNXN PMT POWI PRI PRO QRVO QNST RPD RSG SGEN SKX SWI SYNA TDC TBI USX VRSN VVI VSAT VRTU VCRA WTS WU ZAYO
Friday (Feb 8)
Tuesday, October 24, 2017
=Alexion Pharma (ALXN) receives FDA approval for Soliris
Alexion Pharma receives FDA approval for Soliris
- Co announced the FDA approved Soliris (eculizumab) as a treatment for adult patients with generalized myasthenia gravis who are anti-acetylcholine receptor antibody-positive.
- In the Phase 3 REGAIN study and its ongoing open-label extension study, Soliris demonstrated treatment benefits for patients with anti-AchR antibody-positive gMG who had previously failed immunosuppressive treatment and continued to suffer from significant unresolved disease symptoms, which can include difficulties seeing, walking, talking, swallowing and breathing.
- These patients are at an increased risk of disease exacerbations and crises that may require hospitalization and intensive care and may be life-threatening.
- These patients represent approximately 5-10% of all patients with MG.
Labels:
ALXN,
FDA approval
Monday, October 23, 2017
Thursday, July 27, 2017
=Alexion Pharma (ALXN) reported earnings on Thur 27 July 2017 (b/o)
Alexion Pharma beats by $0.30, beats on revs; raises FY17 EPS above consensus :
- Reports Q2 (Jun) earnings of $1.56 per share, $0.30 better than the Capital IQ Consensus of $1.26; revenues rose 21.1% year/year to $912 mln vs the $846.15 mln Capital IQ Consensus.
- Co raises guidance for FY17, sees EPS of $5.40-5.55 vs. $5.33 Capital IQ Consensus Estimate, compared to $5.10-5.30 prior
- Co raises guidance for FY17 - sees revs of $3.45-3.53 bln vs. $3.47 bln Capital IQ Consensus Estimate, compared to $3.4-3.5 bln prior
Subscribe to:
Posts (Atom)